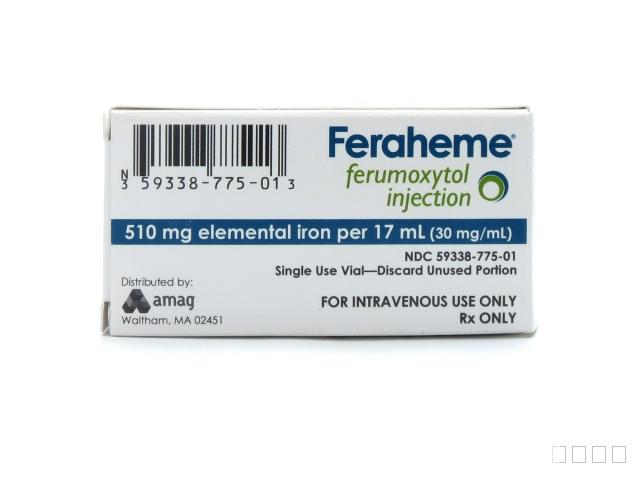
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
药品名:Promethazine 25mg/ml 1ml SDA - Box/25生产商:X-GENProduct Code: CP-5525-3NDC Code: 39822-5525-3
产品名称:CETIRIZINE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:78870持证商:TORRENTNDC:说明书:批准信:
产品名称:METHYLPHENIDATE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:79031持证商:BARR LABS INCNDC:0093-5346-01,0093-5347-01,0093-5348-01说明书:批准信:
产品名称:METHYLPHENIDATE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:79031持证商:BARR LABS INCNDC:0093-5346-01,0093-5347-01,0093-5348-01说明书:批准信:
产品名称:METHYLPHENIDATE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:79031持证商:BARR LABS INCNDC:0093-5346-01,0093-5347-01,0093-5348-01说明书:批准信:
产品名称:METHYLPHENIDATE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:79031持证商:BARR LABS INCNDC:0093-5346-01,0093-5347-01,0093-5348-01说明书:批准信:
产品名称:METHYLPHENIDATE HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:79031持证商:BARR LABS INCNDC:0093-5346-01,0093-5347-01,0093-5348-01说明书:批准信:
产品名称:RIBAVIRIN上市时间:04/19/2023NDA/ANDA/BLA:79111持证商:AUROBINDO PHARMANDC:65862-207-68,65862-207-05说明书:批准信:
产品名称:LAMIVUDINE AND ZIDOVUDINE上市时间:04/19/2023NDA/ANDA/BLA:79124持证商:HETERO LABS LTD IIINDC:43063-900-06,68071-4450-6,71335-0639-2,71335-0639-1,71335-0639-3,31722-506-05,31722-506-60说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2008/079124s000ltr.pdf
产品名称:ONDANSETRON HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:90116持证商:CHARTWELL MOLECULARNDC:说明书:批准信:
产品名称:ONDANSETRON HYDROCHLORIDE上市时间:04/19/2023NDA/ANDA/BLA:90116持证商:CHARTWELL MOLECULARNDC:说明书:批准信: