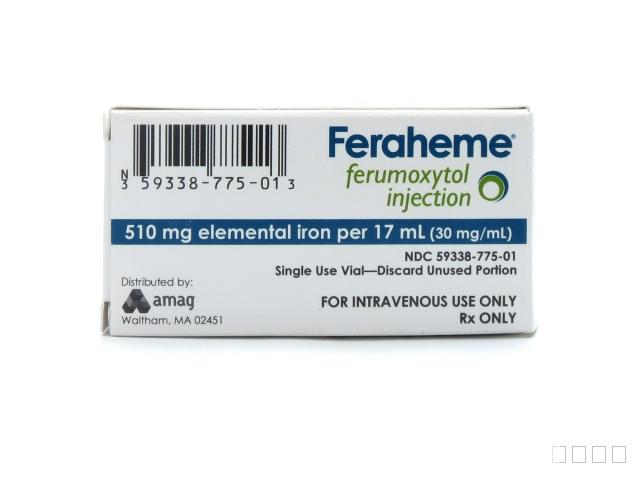
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
药品名:Promethazine 25mg/ml 1ml SDA - Box/25生产商:X-GENProduct Code: CP-5525-3NDC Code: 39822-5525-3
产品名称:BOSENTAN上市时间:02/13/2023NDA/ANDA/BLA:213981持证商:ZYDUS PHARMS USA INCNDC:说明书:批准信:
产品名称:LEVONORGESTREL AND ETHINYL ESTRADIOL上市时间:02/13/2023NDA/ANDA/BLA:212298持证商:HETERO LABSNDC:31722-944-32说明书:批准信:
产品名称:GEFITINIB上市时间:02/13/2023NDA/ANDA/BLA:211591持证商:QILU PHARM HAINANNDC:50742-366-30,67184-0531-1,67184-0531-2,67184-0531-3说明书:批准信:
产品名称:CANAGLIFLOZIN上市时间:02/13/2023NDA/ANDA/BLA:210541持证商:ZYDUS PHARMS USA INCNDC:说明书:批准信:
产品名称:IBRUTINIB上市时间:02/10/2023NDA/ANDA/BLA:215464持证商:SANDOZ INCNDC:说明书:批准信:
产品名称:SILDENAFIL CITRATE上市时间:02/10/2023NDA/ANDA/BLA:212260持证商:NOVITIUM PHARMANDC:70954-168-10说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2019/212260Orig1s000ltr.pdf
产品名称:ZEMDRI 上市时间:02/10/2023NDA/ANDA/BLA:210303持证商:CIPLA USANDC:80735-820-96,71045-010-02,69097-820-96说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210303Orig1s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/210303Orig1s000Ltr.pdf
产品名称:CABAZITAXEL上市时间:02/10/2023NDA/ANDA/BLA:207736持证商:APOTEXNDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/207736Orig1s000TAltr.pdf
产品名称:CABAZITAXEL上市时间:02/10/2023NDA/ANDA/BLA:207718持证商:DR REDDYSNDC:说明书:批准信:
产品名称:ARIKAYCE KIT 上市时间:02/10/2023NDA/ANDA/BLA:207356持证商:INSMED INCNDC:71558-590-28说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/207356s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/207356Orig1s000ltr.pdf