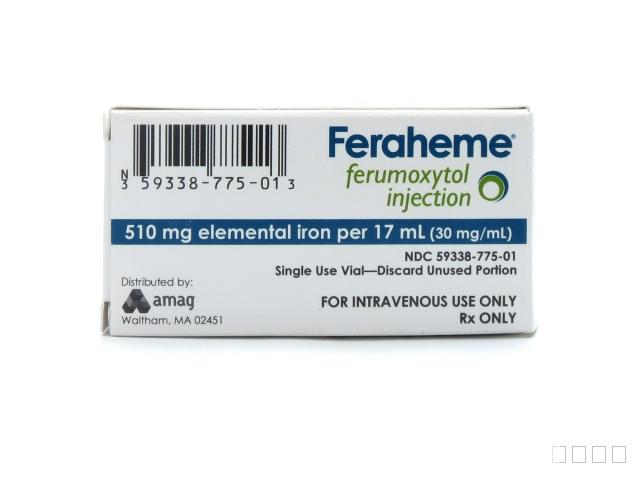
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
药品编号 13748591 持证商 URSAPHARM Arzneimittel GmbH 包装规格 10 ml 产品名 Pollival 0,5mg/ml 剂型 Augentropfen Monopräparat ja 活性物质 Azelastin hydrochlorid 需要医生开处方 nein Apothekenpflichtig ja 信息来源:https://www.besamex.de/product/pollival-0-5-mg-ml-augentropfen-loesung.645941.html
产品名称:VIMPAT上市时间:04/28/2023NDA/ANDA/BLA:22254持证商:UCB INCNDC:0131-1810-67说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/022254lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2008/022253s000, 022254s000ltr.pdf
产品名称:VIMPAT上市时间:04/28/2023NDA/ANDA/BLA:22253持证商:UCB INCNDC:0131-2477-35,0131-2477-60,0131-2477-99,0131-2478-35,0131-2478-60,0131-2478-99,0131-2479-35,0131-2479-60,0131-2479-99,0131-2480-35,0131-2480-60,0131-2480-99说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/022253lbl.pdf批准信:https:/...
产品名称:DOXYCYCLINE上市时间:04/27/2023NDA/ANDA/BLA:217674持证商:ALEMBIC PHARMS LTDNDC:说明书:批准信:
产品名称:GUANFACINE HYDROCHLORIDE上市时间:04/27/2023NDA/ANDA/BLA:217293持证商:AJANTA PHARMA LTDNDC:27241-242-01,27241-243-01说明书:https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=830c0be8-2137-46ee-94c6-aa048b7de35f&type=pdf批准信:
产品名称:ABILIFY ASIMTUFII上市时间:04/27/2023NDA/ANDA/BLA:217006持证商:OTSUKANDC:59148-102-80,59148-114-80说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/217006s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/217006Orig1s000ltr.pdf
产品名称:CHLORZOXAZONE上市时间:04/27/2023NDA/ANDA/BLA:214365持证商:ACME LABSNDC:说明书:批准信:
产品名称:THALIDOMIDE上市时间:04/27/2023NDA/ANDA/BLA:213267持证商:NATCONDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/213267Orig1s000ltr.pdf
产品名称:THALIDOMIDE上市时间:04/27/2023NDA/ANDA/BLA:213267持证商:NATCONDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/213267Orig1s000ltr.pdf
产品名称:ESOMEPRAZOLE MAGNESIUM上市时间:04/27/2023NDA/ANDA/BLA:211977持证商:HETERO LABS LTD IIINDC:71335-2040-1,71335-2040-2,71335-2040-3,71335-2040-4,71335-2040-5,71335-2040-6,71335-2040-7,68071-2649-9,80425-0331-1,80425-0331-2,80425-0331-3,31722-664-10,31722-664-30,31722-664-90,31722-665-10,31722-665-30,3172...
产品名称:ESOMEPRAZOLE MAGNESIUM上市时间:04/27/2023NDA/ANDA/BLA:211977持证商:HETERO LABS LTD IIINDC:71335-2040-1,71335-2040-2,71335-2040-3,71335-2040-4,71335-2040-5,71335-2040-6,71335-2040-7,68071-2649-9,80425-0331-1,80425-0331-2,80425-0331-3,31722-664-10,31722-664-30,31722-664-90,31722-665-10,31722-665-30,3172...