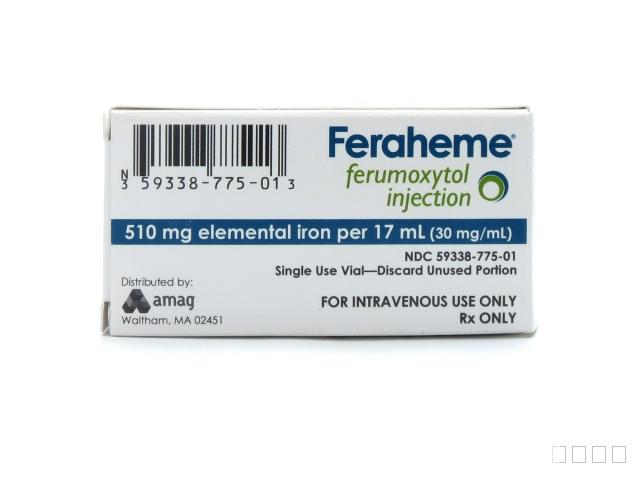
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
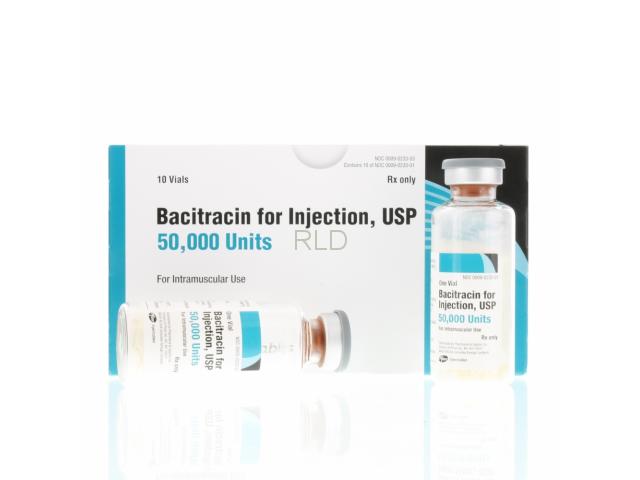
药品名:Bacitracin for Injection 50,000 Units Per Vial, USP - Box/10Product Code: CP-07-0023303NDC Code: 0009-0233-03生产商:Pfizer
产品名称:DICHLORPHENAMIDE上市时间:12/29/2022NDA/ANDA/BLA:215924持证商:TORRENTNDC:13668-676-01,13668-676-30说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/215924Orig1s000ltr.pdf
产品名称:CANAGLIFLOZIN; METFORMIN HYDROCHLORIDE上市时间:02/16/2023NDA/ANDA/BLA:210542持证商:ZYDUS PHARMS USA INCNDC:说明书:批准信:
产品名称:DORZOLAMIDE HYDROCHLORIDE AND TIMOLOL MALEATE上市时间:03/08/2023NDA/ANDA/BLA:203058持证商:AKORNNDC:说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/203058Orig1s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2014/203058Orig1s000ltr.pdf
产品名称:ZIPRASIDONE HYDROCHLORIDE上市时间:03/30/2023NDA/ANDA/BLA:77560持证商:LUPIN PHARMSNDC:68180-331-07,68180-332-07,70518-2622-0,68180-333-07,68180-334-07说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2007/077560_TA_ltr.pdf
产品名称:AZITHROMYCIN上市时间:07/14/2022NDA/ANDA/BLA:205666持证商:AMNEALNDC:65162-708-83,65162-759-39,65162-758-83,65162-760-84说明书:批准信:
产品名称:DARUNAVIR上市时间:08/24/2022NDA/ANDA/BLA:215389持证商:MSN LABS PVT LTDNDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/215389Orig1s000TAltr.pdf
产品名称:ZOLPIDEM TARTRATE上市时间:09/28/2022NDA/ANDA/BLA:77884持证商:APOTEX INCNDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2007/077884s000ltr.pdf
产品名称:LIFITEGRAST上市时间:11/10/2022NDA/ANDA/BLA:215081持证商:MICRO LABS USANDC:说明书:批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/215081Orig1s000TAltr.pdf
产品名称:POTASSIUM CHLORIDE上市时间:12/30/2022NDA/ANDA/BLA:214892持证商:GUARDIAN DRUGNDC:53041-680-06,53041-681-06,53041-681-17,53041-681-29说明书:批准信:
产品名称:LAMZEDEBLA 上市时间:02/16/2023NDA/ANDA/BLA:761278持证商:CHIESI FARMACEUTICI SPANDC:10122-180-02,10122-180-05,10122-180-10说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761278s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/761278Orig1s000ltr.pdf