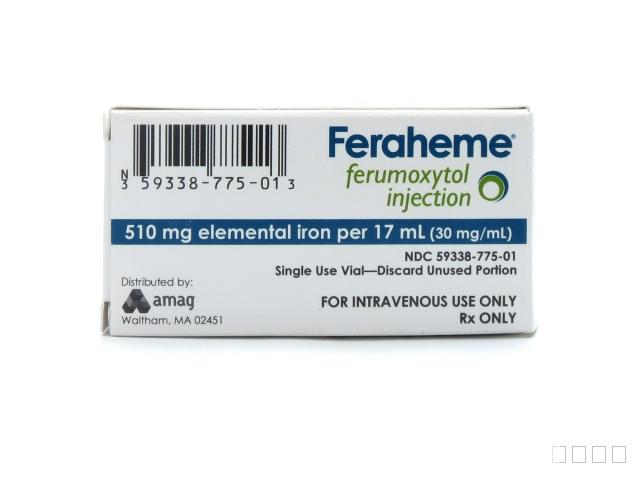
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
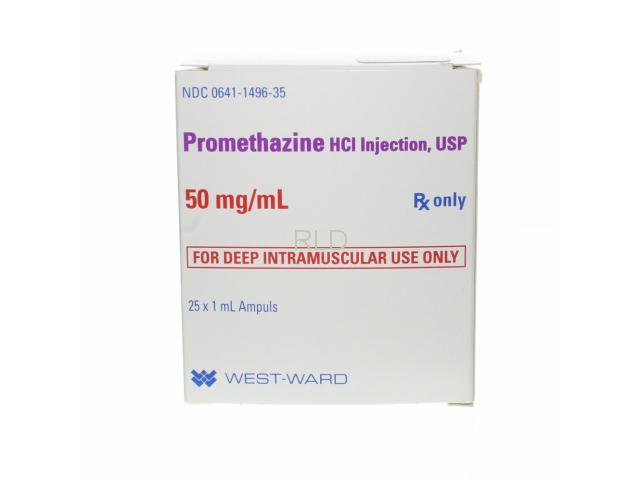
药品名:Promethazine 50mg/ml 1ml SDA - Box/25生产商:WEST-WARDProduct Code: CP-07-0149635NDC Code: 0641-1496-35
产品名称:SAXEND上市时间:04/20/2023NDA/ANDA/BLA:206321持证商:NOVONDC:0169-2800-15,0169-2800-90,0169-2800-97说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/206321Orig1s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2014/206321Orig1s000ltr.pdf
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:208947持证商:NOVAST LABSNDC:50742-635-10,50742-638-10,50742-639-10,50742-639-30,34908-636-10,34908-636-30,34908-636-60,34908-636-90,34908-637-10,34908-637-30,34908-637-60,34908-637-90,34908-638-10,34908-638-30,34908-638-60,34908-638-90,34908-639-10,34...
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:208947持证商:NOVAST LABSNDC:50742-635-10,50742-638-10,50742-639-10,50742-639-30,34908-636-10,34908-636-30,34908-636-60,34908-636-90,34908-637-10,34908-637-30,34908-637-60,34908-637-90,34908-638-10,34908-638-30,34908-638-60,34908-638-90,34908-639-10,34...
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:208947持证商:NOVAST LABSNDC:50742-635-10,50742-638-10,50742-639-10,50742-639-30,34908-636-10,34908-636-30,34908-636-60,34908-636-90,34908-637-10,34908-637-30,34908-637-60,34908-637-90,34908-638-10,34908-638-30,34908-638-60,34908-638-90,34908-639-10,34...
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信:
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信:
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信:
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信:
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信:
产品名称:QUETIAPINE FUMARATE上市时间:04/20/2023NDA/ANDA/BLA:211405持证商:AMNEAL PHARMSNDC:65162-431-06说明书:批准信: