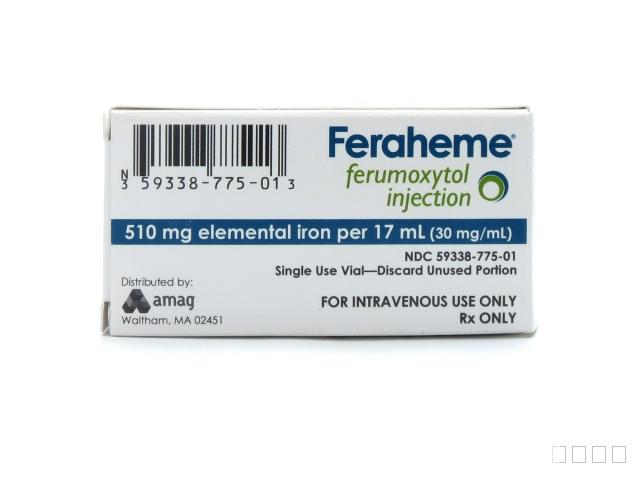
NDC:59338-775-01活性物质:FERUMOXYTOL剂量:510 mg/ 17 mL服用方式:静脉注射NDA:NDA022180 首次上市时间:07/13/2009分销商:AMAG Pharmaceuticals, Inc.生产商一:Baxter Pharmaceutical Solutions生产商二:Patheon Manufacturing Services LLC说明书:PDF
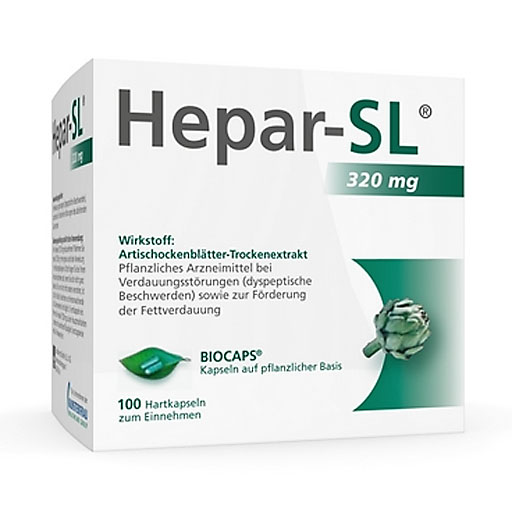
药品编号 09530449 持证商 MCM KLOSTERFRAU Vertr. GmbH 包装规格 100 St 剂型 Hartkapseln 需要医生开处方 nein Apothekenpflichtig nein 信息来源:https://www.besamex.de/produkt/hepar-sl-320-mg-hartkapseln-100-st-09530449.html
Pregabalin Powder CAS No.148553-50-8
5mg 10mg 15mg Vial, 10 vials per box
Lose Weights Peptide 5mg 10mg 15mg Vial(Semaglutide /Tirzepatide), 10 vials one box
5mg, 10mg, 15mg Vial, 10 vials per box/ 1g, 5g, 10g bottle. My email is toppharm1999@gmail.com
5mg 10mg 15mg Vial, 10 vials per box. My email is toppharm1999@gmail.com
5mg 10mg 15mg Vial Semaglutide Tirzepatide, 10 Vials per box. My email is toppharm1999@gmail.com
产品名称:SOGROYA上市时间:04/28/2023NDA/ANDA/BLA:761156持证商:NOVO NORDISK INCNDC:0169-2030-11,0169-2030-90,0169-2035-11,0169-2035-90,0169-2037-11,0169-2037-90说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761156s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2020/761156Or...
产品名称:LIBTAYO上市时间:04/28/2023NDA/ANDA/BLA:761097持证商:REGENERON PHARMACEUTICALSNDC:61755-008-01说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761097s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/761097Orig1s000ltr.pdf
产品名称:LIBTAYO上市时间:04/28/2023NDA/ANDA/BLA:761097持证商:REGENERON PHARMACEUTICALSNDC:61755-008-01说明书:https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761097s000lbl.pdf批准信:https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/761097Orig1s000ltr.pdf
产品名称:ESTRADIOL VALERATE上市时间:04/28/2023NDA/ANDA/BLA:216656持证商:XIROMEDNDC:70700-273-22,70700-274-22,70700-275-22说明书:https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=2e5c7559-7420-168d-6272-92bb4d20035d&type=pdf批准信: